Use of First-Line PARP Inhibitors in Ovarian Cancer | Published in healthbook TIMES Oncology Hematology

Niraparib: A Review in First-Line Maintenance Therapy in Advanced Ovarian Cancer | Targeted Oncology

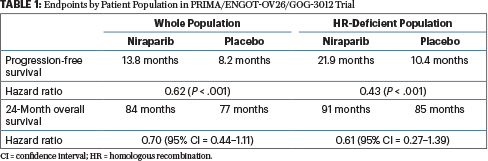
Progression-free survival and safety at 3.5 years of follow-up: results from the randomised phase 3 PRIMA/ENGOT-OV26/GOG-3012 trial of niraparib maintenance treatment in patients with newly diagnosed ovarian cancer - European Journal of

Efficacy and safety of niraparib as maintenance treatment in patients with newly diagnosed advanced ovarian cancer using an individualized starting dose (PRIME Study): A randomized, double-blind, placebo-controlled, phase 3 trial (LBA 5) -

Safety and management of niraparib monotherapy in ovarian cancer clinical trials | International Journal of Gynecologic Cancer

Stephen V Liu, MD on X: "#ESMO19 Practice changing study presented by Antonio Gonzalez Martin. PRIMA study of niraparib in patients with ovarian cancer responding to 1L platinum based chemo. Primary endpoint

Quality-adjusted time without symptoms of disease or toxicity and quality-adjusted progression-free survival with niraparib maintenance in first-line ovarian cancer in the PRIMA trial - Maria-Pilar Barretina-Ginesta, Bradley J. Monk, Sileny Han, Bhavana
Niraparib Efficacy and Safety in Patients with BRCA-Mutated (BRCAm) Ovarian Cancer: Results from Three Phase 3 Niraparib Trials

Quality-adjusted time without symptoms of disease or toxicity and quality-adjusted progression-free survival with niraparib maintenance in first-line ovarian cancer in the PRIMA trial

Progression-free survival and safety at 3.5 years of follow-up: results from the randomised phase 3 PRIMA/ENGOT-OV26/GOG-3012 trial of niraparib maintenance treatment in patients with newly diagnosed ovarian cancer - ScienceDirect

Stephen V Liu, MD on X: "#ESMO19 Practice changing study presented by Antonio Gonzalez Martin. PRIMA study of niraparib in patients with ovarian cancer responding to 1L platinum based chemo. Primary endpoint
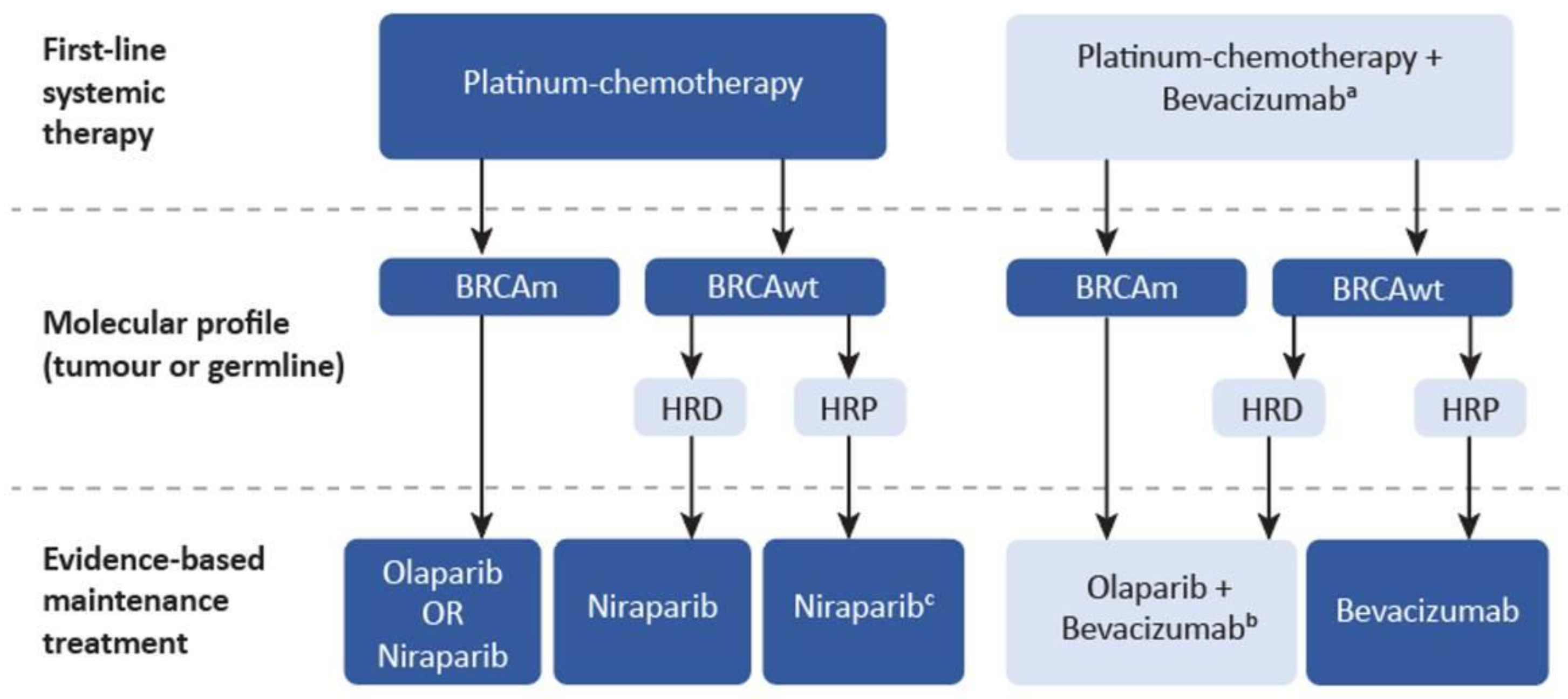
Current Oncology | Free Full-Text | A Pan-Canadian Consensus Statement on First-Line PARP Inhibitor Maintenance for Advanced, High-Grade Serous and Endometrioid Tubal, Ovarian, and Primary Peritoneal Cancers

Prospective evaluation of the tolerability and efficacy of niraparib dosing based on baseline body weight and platelet count : results from the PRIMA/ENGOT-OV26/GOG-3012 trial